LOINC ® and SNOMED CT ® - Why they're Better Together.
To improve interoperability in healthcare it's important to consider coding standards. For clinical measurements and observations can we use just one?

In the last post we took a brief look at some of the coding standards used to identify diagnosis and procedures. This week, I'd like to consider a couple of code sets that represent clinical measurements and observations.
Logical Observation Identifiers Names and Codes (LOINC ®)
The LOINC code set was created in response to a growing desire to electronically exchange clinical data. Development was initiated in the mid 1990's at the Regenstrief Institute, a US-based medical research organization. Clem J. McDonald, MD. led the team in their focus on the exchange of clinical laboratory data. The code set has since expanded. It now contains both laboratory and clinical content. Including observations that relate to both individuals, populations, and the environment. (E.g. vital signs, document titles, patient outcomes, etc.)
The use of this code set allows organizations to create meaningful labels within their systems. Without sacrificing the ability to exchange data with others. As long each organization maps a data element to the same LOINC code, exchange is possible. Each organization is able to have a different display names within their application, if desired. For example:
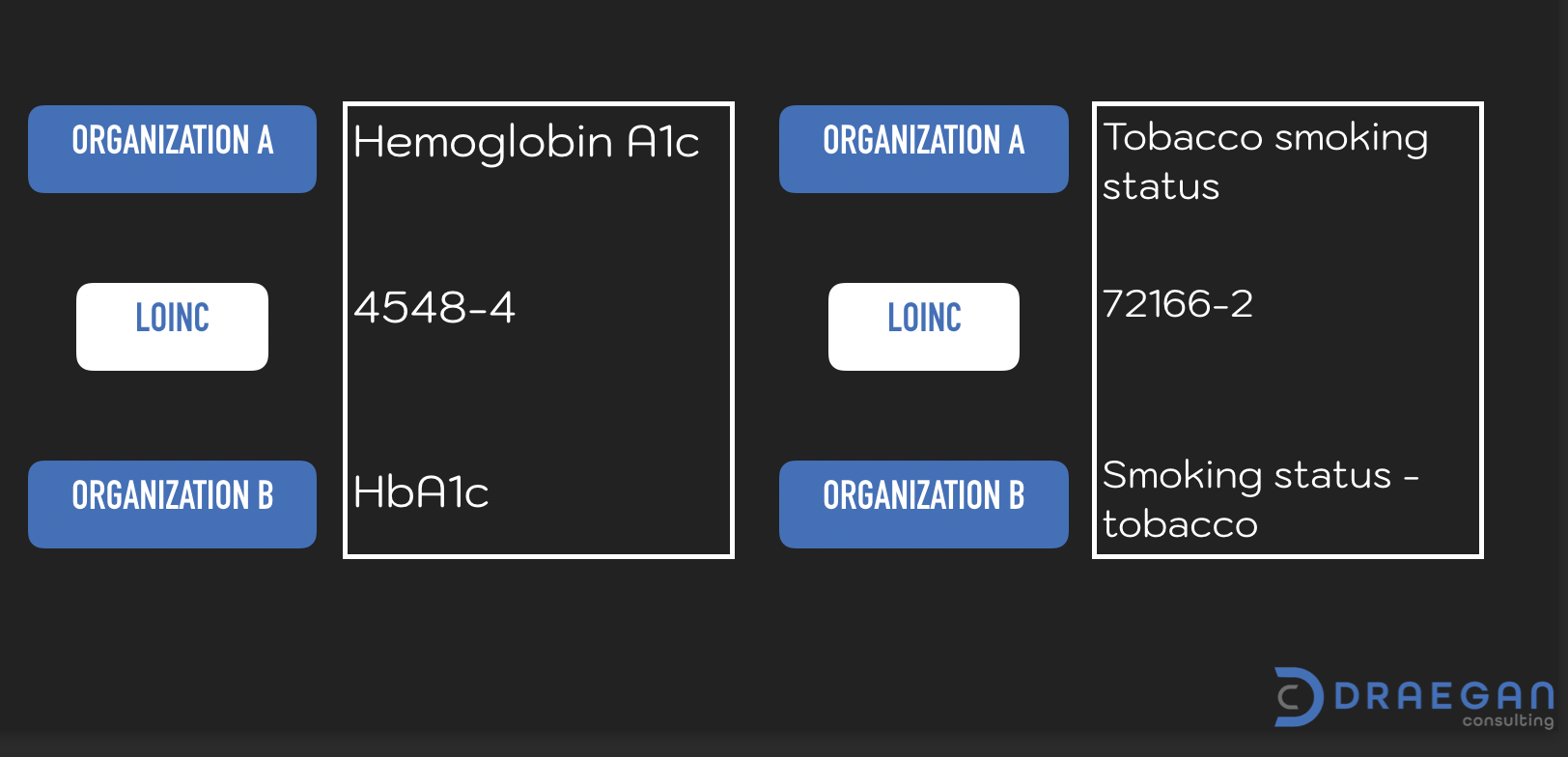
As you can appreciate, the mapping of LOINC codes to an item catalog is not a simple task. One must review each item carefully to associate the correct code value. This requires an intimate familiarity with each of the items. A mapping tool known as RELMA (Regenstrief LOINC Mapping Assistant) is available to assist organizations. Though it is important to note that use of this tool still requires individuals make the final determinations.
In 1999, the standards organization Health Level 7 (HL7), named LOINC as their preferred code set for Lab test data transactions. Then, in 2009, the US Meaningful Use program included the use of LOINC as a required standard for several measures. (This program, now called 'Promoting Interoperability, continues to require the standard.) LOINC is in use by 176 countries around the world; and, of those is mandated by approximately 30. The Regenstrief Institute continues to maintain the data set. With updates biannually in June and December. The purpose of this code is described perfectly on the LOINC.org website:

Pan-Canadian LOINC Observation Code Database (pCLOCD)
Canada, like many other countries, maintains a targeted subset of LOINC codes approved for use within the country’s healthcare industry. To assist with mapping efforts, the Regenstrief Institute is able to upload a country’s data subset to the RELMA tool. This allows users to search within their respective subset to minimize areas where there may is potential for confusion. Canada Health Infoway is responsible for maintaining pCLOCD. The data set is updated biannually with releases at the end of January and July respectively.
Systemized Nomenclature of Medicine - Clinical Terms (SNOMED CT ®)
SNOMED CT is currently used as a common language for health terms by more than 80 countries. It represents the largest clinical code set in the world. The version in use today is the result of a merger of two previous code sets. The SNOMED Reference Terminology (SNOMED RT) and the ‘Read Codes’ developed by the UK National Health Service (NHS). Content is broken down into three main components: concepts, descriptions, and relationships. Used together, these combine to standardize clinical phrases.
SNOMED CT codes are available for many health terms. These include, but are not limited to:
- diagnosis and procedures
- symptoms
- family history
- allergies
- observations
- devices
- assessment tools
The International Health Terminology Standards Organization (IHTSO) owns the standard. However, a non-profit organization called SNOMED International handles maintenance.
Within the US, the use of SNOMED CT is required with the Promoting Interoperability program as well as several other reporting programs throughout the country. Similar to other code sets, many countries develop subsets for use within their health systems. (The Canadian subset is developed and maintained by Canada Health Infoway )
Building on the LOINC smoking example, the following SNOMED CT codes represent some of the possible ‘answers’ to the Tobacco smoking status ‘question’.

Global initiatives for the exchange of health information continue to be a work in progress. With so many options, it is difficult to predict which standards will become true world-wide standards in the coming years. It can also be hard to decide where to being when codifying an electronic health record (EHR) database. Deciding which code sets to configure will depend on several things. Regulatory requirements, the proliferation of overall use within relevant exchange areas, and need. it is also important to note that for many organizations, it is not unusual to have many coding standards attached to some items.
Until next time ...


